The Periodic Law Part 2 Review Questions and Answers
What is Modern Periodic Police force?
The modern periodic table is adult subsequently the periodic police and a periodic table given past Mendeleev. In the latter function of the 18th century, Mendeleev made his periodic tabular array. Scientists did not know virtually the internal structure of the atom dorsum and then.
The development of various diminutive models and advances in quantum theory revealed that the atomic number is the well-nigh basic property of a chemical element. This led to the modification of Mendeleev's periodic law, which is today chosen as mod periodic law.
Check ⇒ History of Periodic Table
Modern Periodic Law Definition
The modern Periodic law tin be stated as:
"The physical and chemic properties of the elements are periodic functions of their diminutive numbers".
The atomic number is equal to the number of electrons or protons in a neutral atom. After knowing the primal unit of measurement of elements, scientists now had a clear idea well-nigh breakthrough numbers and electronic configuration of elements in the periodic table. After knowing the periodic police force, chemists noticed that there is an analogy between the 94 naturally occurring chemical elements. This analogy made people more than curious about the chemistry of these elements. Scientists fabricated various artificial elements. A new periodic table was adult based on the modern periodic law by modifying the Mendeleev'due south periodic table.
Recommended Videos

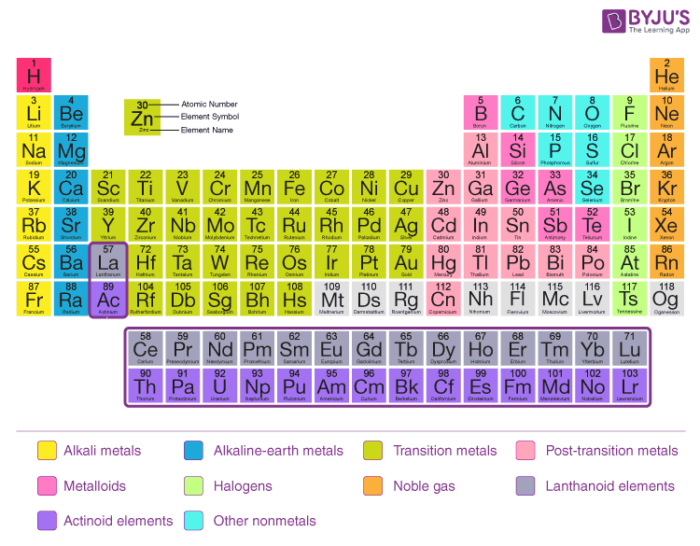
The Modern Periodic Table
The present form of a periodic table that is widely used beyond the earth is the long class of the periodic table. In this form of a periodic table, the horizontal rows are called periods and the vertical columns are known as the groups.
Groups consist of elements that accept similar outer crush electronic configuration in their atoms. Previously the groups were named as IA,…VIIIA, Viii, IB…VIIB and 0. But now they are named equally 1, 2, iii…18. In the modern periodic table, periods are the 7 horizontal rows. Principle quantum number 'n' decides the flow of the element. Principle quantum number (n) is one of the four quantum numbers (n, fifty, thou, and s). It tells usa nigh the principle electron trounce. For instance, if n= iii, then it indicates the principle vanquish every bit 3.
Classification of the Elements in the Periodic Tabular array:
Classification of the elements in the periodic tabular array tin can be done in four ways on the footing of their electronic configurations:
-
Noble gas elements:
Elements of group eighteen of the modern periodic table are considered a noble gas. The electronic configuration of the first element (helium) of this group is 1s2. Balance all the elements (neon, argon, krypton, xenon, and radon) take their outer shell electronic configuration is ns2np6. As the octet of these elements is complete, hence they are highly stable elements.
-
Representative elements:
Southward-lock and P-block elements come under the category of representative elements. Elements in groups 1 and two are known as the due south – block elements (elements with 1s2and 2s2 outermost configuration). Group 13-17 are known every bit the p-block elements (outermost configuration varies from ns2np1 to ns2np5).
-
Transition elements:
Elements which vest to grouping three to 12 and have their outer crush electronic configuration as (northward-ane)d1-10ns1-2 are referred to equally transition elements. These elements are besides known as the d-cake elements.
-
Inner transition elements:
Lanthanides and actinides series which falls at the bottom of the periodic table comes under the category of inner transition elements. In these elements the 4f and 5f orbitals are partially filled, rendering them special backdrop.
Oft Asked Questions
Who gave the modern periodic law?
In 1869, Dmitri Mendeleev and Lothar Meyer established the periodic police force independently. The first periodic table was developed by Mendeleev and soon followed by Meyer. Each grouped the elements by their mass and proposed regularly reoccurring those backdrop.
Why is the periodic law important?
Periodic law is recognized as one of chemistry's most of import concepts. While dealing with the chemic elements, their backdrop, and their chemical reactions, each chemist makes use of Periodic Law, whether consciously or not. The development of the modern periodic table was driven by periodic police.
How do periodic trends relate to periodic law?
Periodic trends are common patterns in the periodic table showing usa the diverse aspects of an element such as electronegativity, diminutive radius, or ionizing power. The periodic police force tells us that when grouped past atomic number, certain properties of elements occur periodically.
Is atomic mass a periodic holding?
Generally, nuclear mass decreases from left to right and ever increases from top to lesser. Equally the atomic number has been developed as the footing for organizing the elements on the periodic tabular array, the atomic number volition e'er increase from left to right and height to bottom.
Which is relative periodic belongings?
In a given menstruation, the valence shell electronic configuration of any two elements is not the same. Because of this, elements throughout time have dissimilar chemic backdrop with a periodic gradation from left to right for their physical properties. This is referred to every bit periodic property.
This is just a brief clarification of the periodic tabular array and the classification of elements. To know more about it, register with BYJU'S & download BYJU'Southward – the learning app.
Learn more about Electron Proceeds Enthalpy here:
If y'all yet have doubts regarding the periodic law and would like to acquire nigh periodic table class ten, bank check out our Introduction to the Periodic Table for detailed clarification.
Source: https://byjus.com/chemistry/modern-periodic-table-modern-periodic-law/
0 Response to "The Periodic Law Part 2 Review Questions and Answers"
Postar um comentário